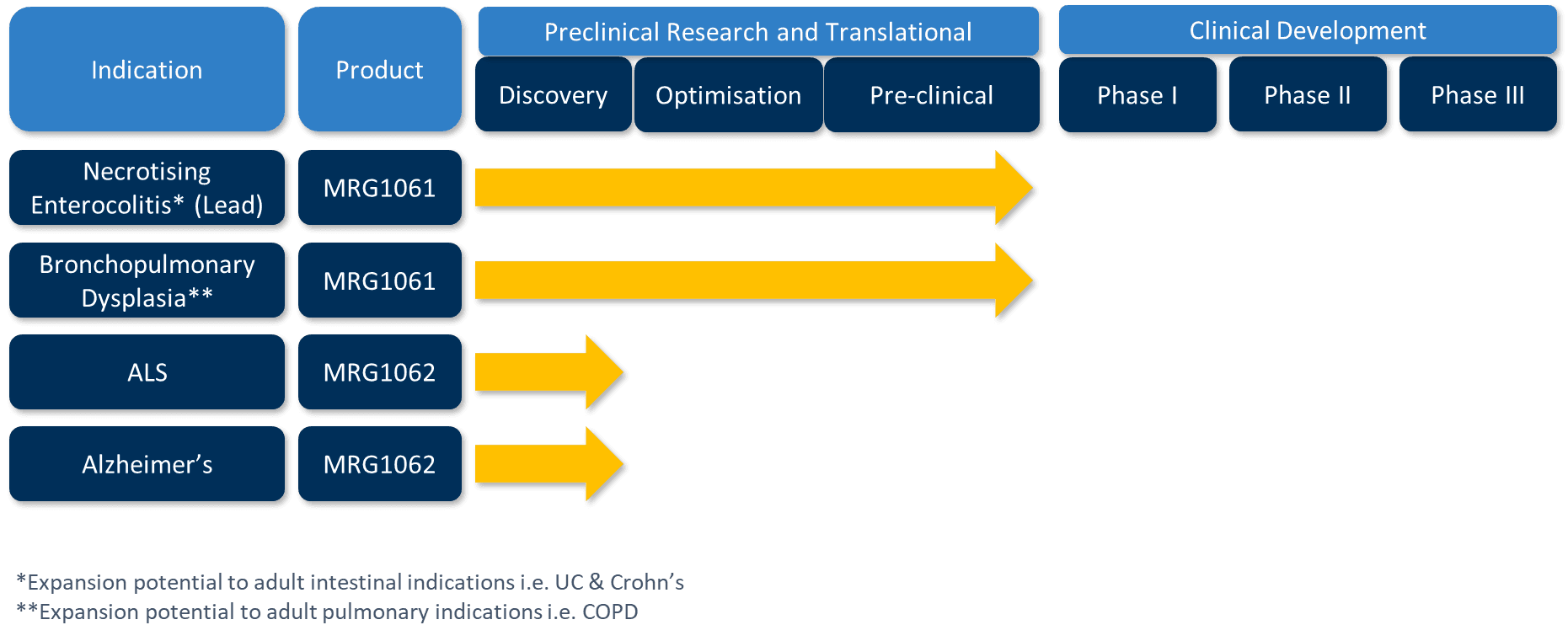
Micregen has strategically chosen to focus its lead therapeutic development on rare neonatal diseases with critical unmet need, such as Necrotising Enterocolitis (NEC) and Bronchopulmonary Dysplasia (BPD). The biology underpinning NEC reveals profound overlap with core mechanisms that underlie a broad spectrum of degenerative and age-associated adult diseases.
The company’s lead Secretomix® candidate, MRG1061, is being advanced based on robust preclinical R&D completed together with world-leading centres (SickKids Hospital, Toronto and Great Ormond Street Hospital, London). Throughout development, guidance from key opinion leaders (KOLs) in neonatology and the regulatory authorities, such as the MHRA, has substantially de-risked the clinical trial pathway, ensuring the programme aligns with the most current scientific and regulatory expectations.
Micregen’s first clinical indications, NEC and BPD, will both be explored in an initial Phase I/IIa trial. These diseases alone represent future markets with forecasted peak annual sales above $1B each, reflecting the scale of unmet need and commercial opportunity.
Beyond neonatal diseases, Micregen’s bioinformatic and AI-based analysis of MRG1061’s secretome cargo has revealed strong links to functional and regenerative pathways relevant to a wide range of age-associated and degenerative diseases. This scientific insight has triggered advanced pipeline programmes targeting conditions such as Motor Neurone Disease and Alzheimer’s, supporting Micregen’s strategy of maximising therapeutic impact and value across diverse indications important in healthy ageing.